With cryogenic condensation, the solvents that are emitted can be efficiently removed from the carrier gas stream without the need for major construction work. Thermal disposal merely recuperates the energy content of the solvents, but the condensation processes offers the possibility of reusing the substances.
Condensation uses a physical process that can be seen in nature in the form of rain, fog or steamy window panes. This process is based on the physical equilibrium between the liquid, solid and gaseous states of a substance. This equilibrium can be roughly described as follows: under stable conditions, the liquid or solid molecules of a substance are converted to the gaseous phase at the same rate at which gaseous molecules condense, or sublime, back to the liquid or solid state. Thus, the concentration of gaseous molecules of the liquid or solid substance in the carrier gas is a result of the vapour pressure which is specific to that substance and which is temperature-dependent.
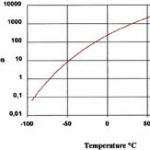
In a two-component system, consisting of a liquid and a gaseous substance, there will, at any given temperature, be a maximum concentration of gaseous molecules of the liquid component in the gaseous substance. This is a function of the partial pressure of the gaseous molecules of the liquid in the mixed gaseous phase. This maximum concentra-tion is called the saturation concentration and is obtained (theoretically) by optimally mixing the gaseous phase above the liquid for an infinite period of time in order to reach a state of equilibrium (Fig. 1).
Principle of indirect condensation
Owing to the temperature dependence of the saturation concentration of volatile substances in a gaseous carrier medium as shown in figure 1, exhaust streams can be cleaned by lowering the temperature so that the solvent starts to condense out.
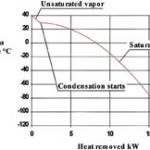
Initially, removing energy from the gas stream causes linear cooling until the temperature is reached at which the actual concentration of the substances to be condensed corresponds to the maximum possible (saturation) concentration. This temperature, at which condensation starts, is called the “dew point”. From this moment on, both the energy required to reduce the temperature and the energy liberated as a result of the substance condensing – which corresponds to the heat of evaporation – has to be withdrawn from the system (Fig. 2).
Therefore, by setting a specific condensation temperature and ensuring optimum separa-tion of the condensate, it is possible to obtain a specific residual concentration of the substance that must be removed from the exhaust stream. However, in order to arrive at the residual values required by the regulatory authorities, it is necessary in many cases to use temperatures that can only be achieved by using liquid nitrogen (LIN) as the coolant. A crucial condition of indirect condensation is that the gas stream to be cleaned does not come into contact with the coolant during the entire process.
Technical implementation
The above principles are implemented using heat exchangers, which, depending on the application, allow different coolants to be combined in a number of cooling units connected in series.
Whilst simple shell-and-tube heat exchangers can be used for conventional coolants, the low temperature of liquid nitrogen (-196 °C at 1 bar) requires specially designed heat exchangers in order to prevent the condensed solvents from freezing. A special feature of the heat exchangers developed for this type of application is that it is possible to control the temperature of the tube walls. This feature allows the use of condensation temperatures that will ensure minimum residual concentrations in the cleaned gas stream, and yet prevent icing inside the heat exchanger. This type of construction results in a particularly flexible system that can respond quickly to variations in the volumetric flow, concentration and temperature of the exhaust stream. In order to make optimum use of the available energy, the Cirrus® Vapour Emission Control system (VEC) from AGA uses the cold gaseous nitrogen (GAN) emitted by the cryogenic condenser and the cool cleaned exhaust stream as coolants in the preceding heat exchangers. This also facilitates multi-stage cooling down to the minimum condensation temperature, so that substances in mixed solvent systems – which might otherwise freeze in the cryogenic condenser – are separated in the preliminary stages.
Fields of application
Indirect condensation requires large amounts of energy, since “cooling energy” is required for both condensation of the solvent and cooling of the carrier medium. The VEC system therefore has a specific field of application that allows optimum results to be achieved with regard to functionality and economic efficiency.
Among the obvious applications for indirect condensation with liquid nitrogen are low-volume exhaust streams that are highly loaded with solvents with a freezing point below -30 °C (Fig. 3). Depending upon its composition, the condensate can be reused either immediately or after distillation.
If the gaseous nitrogen leaving the system can be used again, the costs for emission control are minimal since operating costs are reduced to a minimum. Since the system does not contain any moving parts, main-tenance costs are also minimal. It is this feature which makes the system suitable for cleaning emissions that contain explosive mixtures.
Owing to the low pressure drop across the system, it is ideal for use in circulating systems (such as inert-gas dehydration equipment) or in existing emission control systems, in order to partially clean a highly-charged sub-stream that could cause overloading of the main equipment.
Added flexibility thanks to modular design
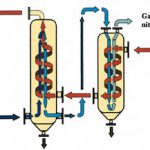
In addition to VEC systems designed to meet the requirements of specific applications, the manufacturer can also supply standard, compact VEC models. The Cirrus M50 module is designed to treat gas flows of up to 50 Nm3 per hour. During the second half of 1998, a new standard module, the Cirrus M150, with a nominal flow capacity of 150 Nm3 per hour, will be launched by this supplier. The standard M50 module consists of two heat exchangers, a cryogenic main condenser and a pre-cooler (Fig. 4).
The pre-cooler uses the cold nitrogen gas emitted by the cryogenic condenser. This allows optimum use to be made of the cooling energy of the nitrogen, and at the same time ensures that substances with a relatively high freezing point are removed by condensation in the pre-cooler, thus reducing the risk of icing in the main cooler. The pre-cooled exhaust stream then enters the main cooler, the cryogenic condenser cooled by liquid nitrogen, where condensation to the desired residual concentration takes place. The main condenser operates in such a way that a fixed condensation temperature is maintained, preventing undesirably low temperatures. The risk of icing in the main cooler is therefore reduced to a minimum. A demister in the outlet of the main cooler prevents condensate droplets from leaving the heat exchanger with the cleaned gas stream. If variations in the composition of the exhaust stream result in icing in the cryogenic condenser, the main cooler can be thawed out quickly using a built-in electrical heating system. The condensation temperature can be adjusted by controlling the flow of liquid nitrogen. The amount of coolant required is proportional to the volume of the gas stream and the amounts of solvents needing to be removed. The standard VEC modules are offered as compact systems installed in a steel frame and equipped with all the necessary control and regulating devices. If the exhaust stream is increased, or if the existing exhaust stream requires a capacity greater than that of the installed module, additional VEC modules can easily be integrated into the system.
Examples of suitable applications for a VEC system include:
l solvent tank vents,
l emission control during filling with solvents,
l processes involving evaporating solvents,
l inert-gas drying and
l distillation equipment.
AGA
Fax: ++49/61/826 72 01
Further information cpp 205
Unsere Whitepaper-Empfehlung
Wasserstoff gilt als Schlüssel für die Dekarbonisierung der Chemieindustrie. Doch die Nutzung des vermeintlichen Hoffnungsträgers Hydrogen birgt auch Gefahren und stellt die Branche vor neue Herausforderungen, die das gratis Whitepaper „H2 wie Hoffnungsträger?“ näher für Sie…
Teilen: